EQUIPOS DE TERAPIA DE INFUSIÓN
Kits de inicio IV, cambio de apósito y cambio de puerto MEDICAL <sup>ACTION</sup>

Kits de inicio IV
Un surtido completo para terapia intravenosa
Los kits de inicio intravenoso de MEDICAL ACTION están diseñados para permitir a los médicos proporcionar una excelente atención al paciente con productos de calidad, al tiempo que se racionalizan los componentes, se reducen los residuos y se ofrecen soluciones de suministro rentables. Los componentes se montan, esterilizan y empaquetan cómodamente para ahorrar un valioso tiempo de enfermería. Al haber menos envases que abrir, el riesgo de contaminación se reduce considerablemente.
- Las opciones de kit incluyen las opciones de apósito 3M™ Tegaderm™ y HALYARD*.
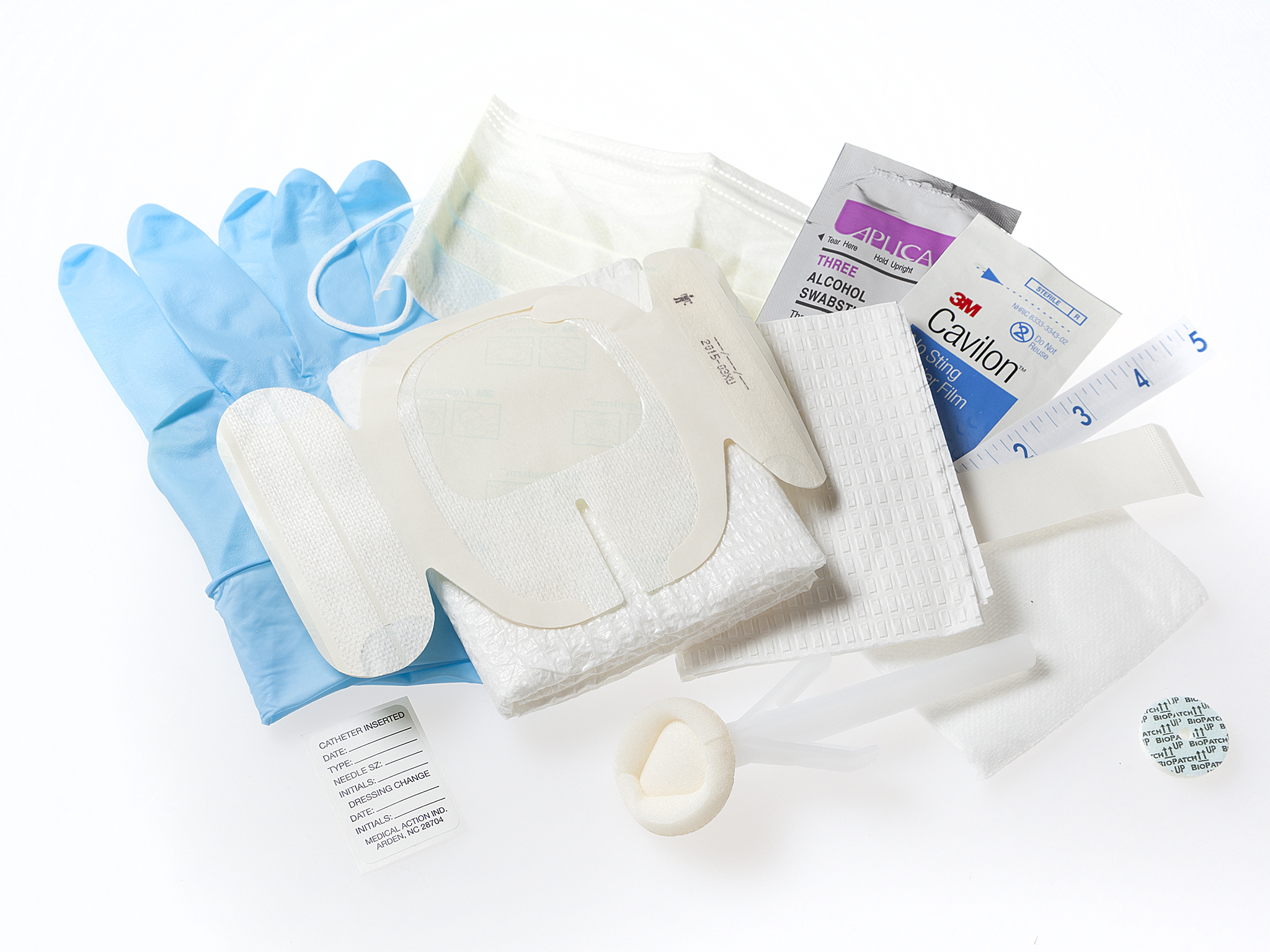
- Los kits incluyen desde guantes, etiquetas, esparadrapo, antisépticos, esponjas de gasa y aplicadores hasta compresas, paños, vendas sin látex, torniquetes y mucho más.
CLEAR SEQUENCE™ Kits de cambio de apósito
Protección del paciente y eficiencia del médico, todo en uno.
Nuestro diseño pendiente de patente separa los pasos de extracción de los de aplicación, reduciendo el riesgo de contaminación. Cada kit CLEAR SEQUENCE está configurado con los componentes específicos y ordenados que requiere su protocolo clínico. El diseño de envoltura única optimiza el uso del espacio de trabajo limitado junto a la cama del paciente
Diseñado a partir de la experiencia clínica, este sistema:
- Organiza los componentes intuitivamente de izquierda a derecha según el protocolo
- Proporciona un campo estéril lo suficientemente flexible como para ser utilizado en una variedad de espacios de trabajo con limitaciones de espacio.
- Ayuda a reducir el riesgo de contaminación microbiana y de otro tipo del emplazamiento

Bolsillos transparentes con instrucciones visuales de uso
- Permite a los clínicos ver cada componente dentro de cada bolsillo
- Ayuda a garantizar que cada paso se sigue según el protocolo
- Reduce el riesgo de que haya elementos que no se vean o pasos que se hayan pasado por alto

Solapa protectora
- Separa los dos pasos principales del procedimiento (retirada y sustitución del apósito) en dos campos estériles independientes.

Elección de componentes
Cada kit CLEAR SEQUENCE™ está configurado con los componentes específicos y ordenados que requiere su protocolo clínico
72.3
Número medio de tareas que realiza una enfermera de quirófano cada hora.
Eso supone 867,6 posibilidades de faltar al protocolo por turno de 12 horas1
Con tantas posibilidades para cada uno, ¿no debería ser cada procedimiento lo más claro posible?
Póngase en contacto con su representante de ventas para obtener más información sobre los kits de cambio de apósitos de secuencia clara de Medical Action.
De más de 100 enfermeras encuestadas
70%
no se sentían muy seguros con el procedimiento de cambio de apósito
Nuestros kits de procedimientos paso a paso CLEAR <sup>SEQUENCE™</sup> se adaptan a su protocolo o formulario específico<br>
para ofrecer a los clínicos la facilidad y eficacia que necesitan.
Con la confianza añadida de CLEAR <sup>SEQUENCE™</sup>, podrá centrarse en el paciente, no solo en el procedimiento.
Kits de acceso a puertos
Los kits de acceso al puerto MEDICAL ACTION® proporcionan todos los componentes necesarios para su procedimiento, incluidos el apósito, el aplicador antiséptico, la mascarilla, los guantes y otros componentes de prevención de infecciones. Elija una de nuestras opciones disponibles o personalice el kit según su protocolo específico de acceso a puertos.
Kits adicionales de cambio de apósitos
Alineándonos con sus objetivos -desde la estandarización en torno a sus protocolos específicos hasta la reducción de costes, la mejora de la continuidad de la enfermería y la incorporación de nuevas tecnologías-, estamos preparados para personalizar kits que respondan a sus retos específicos.
Productos relacionados
¿Tiene preguntas?
Nuestro equipo está dispuesto a ayudarle. Colaboramos con la más amplia gama de fabricantes del sector sanitario y profesionales con formación clínica para producir kits de procedimientos que funcionen como lo hacen sus equipos. Empecemos.
HALYARD - Contact Us
Fuentes
1 BMC Health Serv Res. 2011; 11: 319. Publicado en línea 2011 Nov 24. Doi: 10.1186/1472-6963-11-31

